Research and development work around carbon capture has been on the rise, particularly when it comes to sorbents – the materials tasked with extracting CO2 in a range of advancing carbon capture applications.
As companies work to scale this decarbonization technology, facilities such as the FEECO Innovation Center are providing a pivotal de-risking option for developing the essential aspects of sorbent performance and durability.
Why is Research Around Carbon Capture Increasing?
Capacity for carbon capture and storage (CCS) is anticipated to quadruple by 2030, with cumulative investments in the technology expected to reach USD $80 billion. Alongside a sharp increase in capacity, experts agree that carbon capture is at a turning point.[1]
Growth factors pushing demand are diverse, including:
- Unprecedented demand for energy, particularly from data centers
- Corporate decarbonization mandates
- Net-zero supply-chain requirements
- Carbon pricing and tax credits (45Q, EU ETS, etc.)
Carbon capture and storage is largely accepted as the most promising technology to achieve these goals, especially when it comes to hard-to-decarbonize industries such as cement, iron, and steel. As technologies advance, solid sorbents are shaping up to become a favored approach, but currently represent the primary bottleneck in advancing the technology.
What is a Sorbent?
A sorbent is a material that can capture liquids or gases either through absorption (where material incorporates into the sorbent’s structure) or adsorption (where material adheres to the sorbent’s surface).
Sorbents can be made from a variety of materials and employed in diverse applications. They have long been used in both the industrial and consumer spaces and are perhaps most well known for their use in environmental remediation.
Early carbon capture technology relied on liquid sorbents, specifically liquid amine scrubbing technology, which brought issues such as energy-intensive regeneration, excessive corrosion, and high solvent costs. As these challenges have persisted, solid sorbents have become recognized as a promising alternative.[2]
How Does Solid Sorbent Carbon Capture Work?
The concept of carbon capture with a solid sorbent is simple: the sorbent, a porous material, selectively adsorbs carbon dioxide (CO2) onto its extensive surface area, either from emission sources (post-combustion capture/PCC), or directly from the air (direct air capture/DAC). The CO2 can then be removed (desorbed) from the sorbent and either stored in one of several ways, or put to use in industrial processes. The newly regenerated sorbent can be reused.
With typically lower regeneration energy costs, greater selectivity, and other benefits, solid sorbents hold the potential to improve carbon capture in a number of ways: energy efficiency, reduced environmental impact, and lower operating costs, but challenges remain.[2]
The Need for Better Sorbents
While solid sorbents have proven to be highly effective in lab and pilot settings, they present several challenges that threaten their use at the gigaton scale required.[2]
Sorbent cost can be especially prohibitive, accounting for around 45-65% of total cost in post-combustion capture and 80-90% in direct air capture.[3]
Successful carbon capture lies in the ability of the sorbent to not only capture CO2 from its host gas, but also to maintain its integrity throughout multiple use and regeneration cycles, all at a reasonable cost.
This means that the characteristics of the solid sorbent are crucial to its effectiveness. And as a result, improving sorbents has been a significant focus of research and development work in bringing efficient, low-cost carbon capture to fruition at scale.
Promising Solid Sorbents
Several materials can act as a solid sorbent – activated carbon, zeolites, and more. The two sorbents that seem to be garnering the most attention for carbon capture are metal organic frameworks (MOFs) and calcium oxide (CaO, used in “calcium looping”). Each is being explored for both direct air carbon capture storage (DACCS) and post-combustion point source capture.
Metal Organic Frameworks (MOFs)
Metal organic frameworks, or MOFs, are highly porous, crystalline materials with an exceptional internal surface area. With the capability for changing out both their organic and inorganic structural components, the flexibility of MOFs allows scientists to customize them in almost infinite ways.[2]
The following qualities, for which MOFs are widely recognized, make them especially promising in carbon capture settings:[4]
- Tunable porosity
- Customizable pore chemistry
- Superior gas adsorption and separation capabilities
Research around the development of MOFs uniquely suited to the challenges of carbon capture continues to grow.
Calcium Oxide (CaO)
Calcium oxide (CaO) has long been recognized for its ability to react with carbon dioxide (CO2) to form calcium carbonate (CaCO3). It is also a low-cost, widely available material.
Subsequently, it has been employed in what is known as the calcium looping (CaL) process, in which calcium oxide reacts (i.e., is carbonated) with carbon dioxide (CO2) to form calcium carbonate (CaCO3). The CaCO3 is then processed in a calciner, stripping the CO2 out as its own stream for capture or storage, and leaving the newly regenerated CaO ready for reuse.[5]
While solid calcium looping has yet to reach commercial scale, its effectiveness, paired with advantages such as a small footprint, low energy consumption, and reduced water consumption, continue to see it as a highly explored option.[5]
It’s also important to note that while MOFs represent an exciting, next-generation sorbent, calcium looping is significantly closer to industrial readiness and holds a cost advantage over MOFs, thanks to low-cost, abundant sources of natural limestone.
Developing Successful Solid Sorbents
Among other qualities, producers must be able to produce solid sorbents with a high adsorption capacity, a selectivity for CO2, and sufficient durability to withstand multiple cycles of use and regeneration.
As such, the ability of both metal organic frameworks and calcium oxide (and other materials) to serve as effective sorbents in CO2 depends significantly on their physical and chemical composition.[2]
The FEECO Innovation Center is helping producers to de-risk their processes, reduce scale-up issues, and establish commercial scale-up data. Two key areas of influence in this regard are actively under investigation in the Innovation Center:
Agglomeration
As with some catalysts, agglomeration, or particle size enlargement, of sorbents can bring benefits to the carbon capture and regeneration process.
Carbon capture with a solid sorbent is carried out in a reactor, typically a fixed or fluid bed. The host gas is passed through a packed bed of the solid sorbent, which captures the CO2 through chemical reaction.
As such, an essential aspect of effectively capturing the CO2 in this setting is the surface area available to the gas, or in other words, the extent of gas-to-solid contact. This depends significantly on bed permeability, or the ability of the gas to move through the bed of solids.
Agglomeration helps to improve bed permeability by eliminating fines that could cause clogging and channeling; by binding fines together into larger, more uniform spherical particles, gas is better able to flow through packed sorbent beds. This is illustrated in the image below.
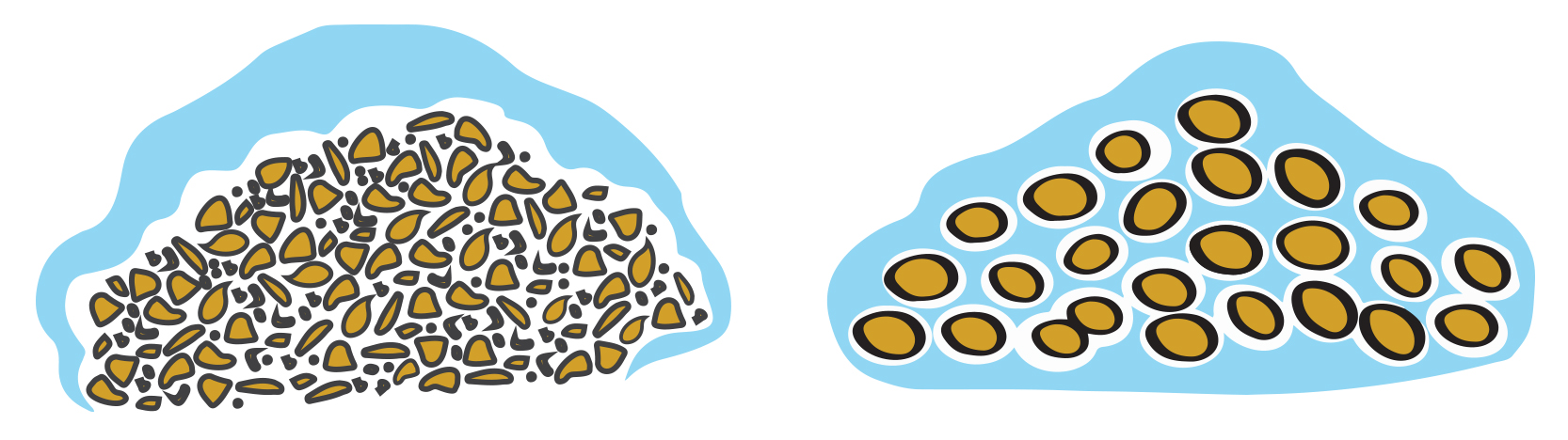
Improved bed permeability through agglomeration of fines
The use of agglomerates over powder also ensures that the sorbent doesn’t become entrained in the process gas and carried out with the exhaust. Agglomeration also makes sorbent handling, transportation, storage, and feeding much more clean and manageable.
In contrast, poorly formed sorbents are not only challenging to handle and feed, but can also increase pressure drop, limit reactor throughput, and drive up sorbent replacement rates—directly impacting capture cost.
As a result, producers are investing significant research into using agglomeration to optimize the characteristics of sorbents.
What Solid Sorbent Properties Can Be Controlled Through Agglomeration?
Agglomeration can be used to control a number of particle qualities, including:
- Particle size distribution
- Bulk density
- Crush strength
- Compression strength
- Moisture content
- Product formulation/chemical composition
- Green strength
- Surface quality
- Surface-area-to-volume ratio (SA:V)
- Porosity
- Flowability
- Attrition
The potential for controlling attrition is of particular interest, as granular sorbents must be strong enough to withstand use in the packed bed, as well as regeneration, for multiple cycles, without breaking down.
All of these properties can be controlled through agglomeration equipment selection and configuration, as well as process variables, all of which can also be tested in the Innovation Center.

Material testing on a disc pelletizer in progress in the Innovation Center
The Innovation Center utilizes batch- and pilot-scale agitation agglomeration (wet granulation/non-pressure agglomeration) equipment such as pin mixers, disc pelletizers, and agglomeration and coating drums to help producers establish product formulations and physical specifications, as well as the processes required to produce them at scale.
Calcination
Calcination, or other types of high-temperature thermal treatment for carbon capture, is also under investigation in the Innovation Center.
When it comes to MOFs and calcium oxide, calcination is used in two key ways:
MOF Production
MOFs are extremely customizable, with producers modifying them in nearly endless ways to suit their objectives. High-temperature thermal treatment such as calcination may be used in the production of the MOF itself, or in the production of its precursors.
The highly custom nature of MOFs requires honing in on the precise combination of process variables needed to yield the desired specifications.
Calcium Oxide Regeneration
In the calcium looping category, calcination is used to regenerate the sorbent, playing a significant role in the viability of the calcium looping process. This requires careful control over process variables in order to ensure maximum regeneration without product degradation.
In both MOFs and CaO, as well as with other types of sorbents, the Innovation Center utilizes batch- and pilot-scale test kilns to generate the design-ready data needed for reactor sizing, material handling, and regeneration system engineering.
Several variables can be tested to achieve desired characteristics:
- Temperature profiles
- Kiln configuration (direct/indirect)
- Airflow configuration (co-current or counter-current – direct-fired kilns only)
- System pressures
- Retention time
- Kiln rotation speed and slope
- Kiln atmosphere
- Internals to modify bed action
- Percent fill

Direct-fired batch kiln used for testing in the Innovation Center
The data gathered during testing in the Innovation Center is then used to inform on commercial equipment design decisions, allowing FEECO to manufacture the custom equipment necessary to build the process at scale.
Conclusion
As carbon capture moves from pilot projects toward gigaton-scale deployment, the performance of solid sorbents is emerging as one of the most decisive factors in whether these systems can succeed commercially. High-capacity materials such as MOFs and calcium oxide offer enormous promise, but their real-world viability depends not just on chemistry, but on whether they can be manufactured, handled, regenerated, and recycled reliably.
This is where process development such as that carried out in the FEECO Innovation Center becomes the critical bridge between laboratory breakthroughs and bankable CCS projects.
Through its Innovation Center, FEECO provides carbon-capture developers with the pilot-scale agglomeration, thermal processing, and materials-handling infrastructure needed to transform experimental sorbents into durable, reactor-ready products. By testing how materials behave under realistic operating conditions—including pellet strength, attrition, regeneration stability, and flow behavior—producers gain the design-ready data needed to scale confidently into commercial systems.
With its combination of process expertise, batch- and pilot-scale equipment, and decades of experience in handling challenging solids, the FEECO Innovation Center is helping ensure that tomorrow’s carbon-capture sorbents are effective, scalable, reliable, and ready for industrial deployment. To learn how the Innovation Center can assist in your solid sorbent development efforts, contact us today!
SOURCES:
- DNV. “Turning Point for CCS Is Now, DNV Report Finds, With Global Capture and Storage Capacity Expected to Quadruple by 2030.” DNV, June 12, 2025, https://www.dnv.com/news/2025/turning-point-for-ccs-is-now-dnv-report-finds-with-global-capture-and-storage-capacity-expected-to-quadruple-by-2030/
- Zentou, H., et al. “Recent Advances and Challenges in Solid Sorbents for CO₂ Capture.” ScienceDirect, 2025, https://www.sciencedirect.com/science/article/pii/S2772656825000260
- “Atoco.” “What Is a Carbon Capture Sorbent?” Atoco, https://atoco.com/blogs/what-is-a-carbon-capture-sorbent/
- Gavrilović, Ljubomir, S.S. Kazi, A. Oliveira, O.L.I. Encinas, and E.A. Blekkan. “Metal-Organic Frameworks for CO₂ Capture: Challenges and Efforts From Laboratory Research to Industrial Applications.” Industrial & Engineering Chemistry Research, vol. 64, 2025, DOI: 10.1021/acs.iecr.4c04997.
- Paulsen, M.M., et al. “Is Solid Calcium Looping a Scalable Technology for Mega-Ton Carbon Dioxide Removal?” ScienceDirect, 2025, https://www.sciencedirect.com/science/article/pii/S2212982025002392

